Caution — INVESTIGATIONAL DEVICEs, LIMITED BY FEDERAL (OR UNITED STATES) LAW TO INVESTIGATIONAL USE. These products have not been approved by the FDA as safe or effective.
Anyone who has glaucoma is likely to desire a treatment other than eye drops. Currently available drops can be expensive, inconvenient, difficult to administer, and associated with bothersome side effects. In an effort to address these issues a number of implants are under development. These implants would contain glaucoma medication that would be slowly released over many months (known as “Sustained Drug Delivery”). If these implants prove to be both safe and effective then much of the hassle associated with glaucoma treatment could be eliminated.
Dr. Donald L. Budenz provided an update regarding Sustained Drug Delivery for Glaucoma Treatment at the recent American Glaucoma Society (AGS) 2017 Annual Meeting. He reviewed the prostaglandin-analog therapies currently in development along with their early results and limitations.
Prostaglandin-Analog Therapies Currently in Development
Punctal Plugs
There is a small opening in each eyelid near the nose. This opening (called the “puncta”) allows tears to drain into the sinuses. For those with dry eyes, these puncta can be closed using punctal plugs. It is possible to create plugs that are imbedded with medication. This medication would be slowly released over many months. Currently there are two such plugs being studied for the potential treatment of glaucoma:
-
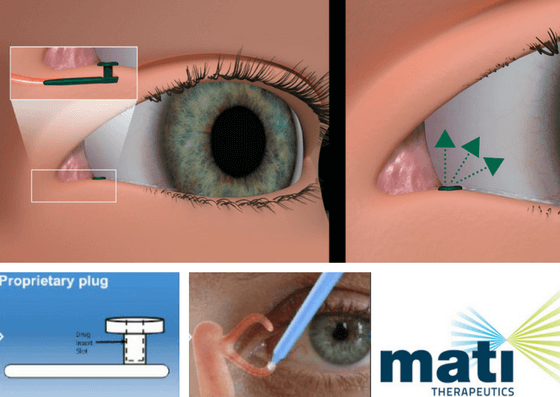
Mati Therapeutics – Latanoprost Plug
This looks just like a plug used for the treatment of dry eye.
-
Ocular Therapeutix – Travoprost Plug
This plug is cylinder-shaped and sits inside the puncta just below the surface of the eyelid.
Issues with plugs:
- They may spontaneously fall out
- They can be associated with tearing
- The skin color may change around the plug
Fornix Ring
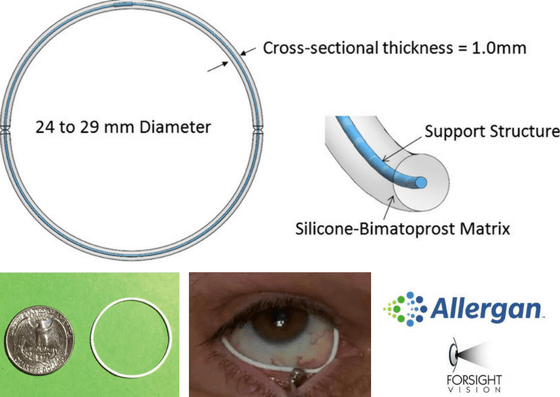
Bimatoprost Ring
This is a flexible ring that is placed under the lid. The ring sits in the fornices (where contact lenses sometimes get “lost”). It seems to be generally well tolerated though a small section of the ring can be seen near the nose where the eyelids come together. Some people have found that to be cosmetically undesirable.
Intraocular Injectable Implants
In addition to placing glaucoma sustained drug delivery implants outside of the eye, it is also possible to inject them into the eye. Such treatments are already available for retinal diseases. Implants currently being evaluated in the treatment of glaucoma would be injected into the front of the eye (anterior chamber). Two such implants are currently in clinical trials:
-
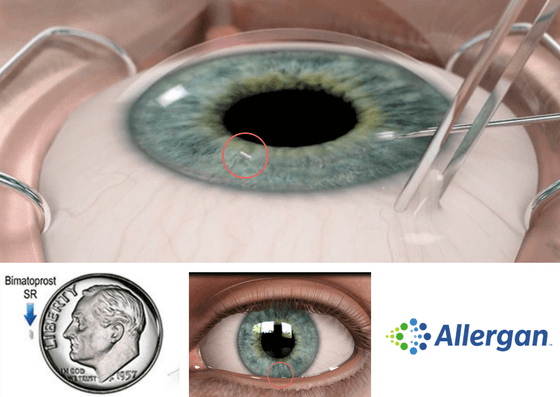
Bimatoprost SR
-
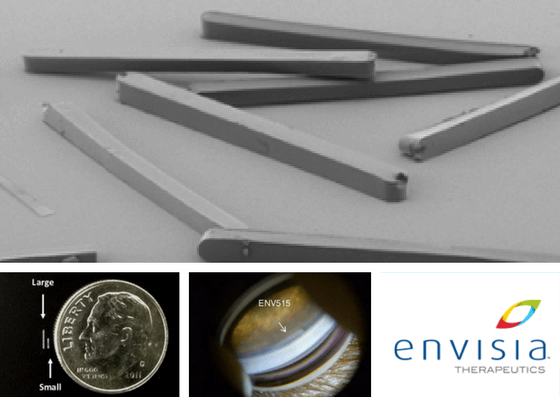
Envisia ENV515 Travoprost XR
Interestingly, this was the only Sustained Drug Delivery implant that did not show signs of “IOP drift” over time.
Issues with injectable sustained drug delivery systems:
- Potential for intraocular inflammation
- Potential for cataract formation
- Potential for infection leading to loss of vision
The above implants all seem to lower IOP as well as eyedrops containing the same medication. However, all but the Envisia EN515 Travoprost XR showed some upward drift of IOP over time.
It is quite likely that one or more of these implants will be FDA approved over the next few years for the treatment of glaucoma. It is also quite likely that they will be expensive. Whether insurance covers the cost of these implants will likely determine whether they are widely used.
Don’t delay getting checked for glaucoma
Make an appointment with an eye doctor in your area now. If you live in the greater Los Angeles area and would like Dr. David Richardson to evaluate your eyes for glaucoma call 626-289-7856 now. Appointments are available Tuesday through Saturday.

David Richardson, MD
Medical Director, San Marino Eye
David Richardson, M.D. is recognized as one of the top cataract and glaucoma surgeons in the US and is among an elite group of glaucoma surgeons in the country performing the highly specialized canaloplasty procedure. Morever, Dr. Richardson is one of only a few surgeons in the greater Los Angeles area that performs MicroPulse P3™ "Cyclophotocoagulation" (MP3) glaucoma laser surgery. Dr. Richardson graduated Magna Cum Laude from the University of Southern California and earned his Medical Degree from Harvard Medical School. He completed his ophthalmology residency at the LAC+USC Medical Center/ Doheny Eye Institute. Dr. Richardson is also an Ambassador of Glaucoma Research Foundation.